Ice storage is sometimes used in central air conditioning systems as a way of smoothing demand for chilling, thereby reducing the installed chiller capacity or allowing demand to be time-shifted. It’s attractive because the latent heat absorbed or released as the water changes phase between liquid and solid is an order of magnitude more than can be stored and recovered just by heating or cooling liquid water.
With phase-change storage established as a legitimate and effective element of central air conditioning and heating plant, it should come as no surprise that we now see vendors offering phase-change materials (PCM) to be embedded in the fabric of buildings as a way of stabilising internal temperatures and thus (according to their claims) saving energy. Are such claims likely to have any merit?
The concept of a PCM such as ice is that as any substance melts (or solidifies), it absorbs (or releases) heat without a change in temperature. PCMs for use in building elements such as walls or ceilings are usually either salts or waxes that change phase at the building’s internal set-point temperature. The argument goes that when daily outside-air temperatures swing above and below the internal set-point, heat stored during the hot part of the day is released during the cold part, avoiding the need for artificial cooling or heating. However, such circumstances are rare. What would happen in a more realistic scenario where, say, the weather is cold and the space needs heating? Firstly, if the space needs heating continuously, the PCM will never change phase and will thus be redundant. It will either be permanently solid or permanently liquid, depending on which side of its melting point the space is being held.
Now suppose the space is heated intermittently. If the internal set-point were below the PCM’s melting point, it would never melt, so it would have no effect. But if the heating set-point were above the PCM’s melting point then it would absorb heat during the warm-up part of the heating cycle. The problem with this is that it would retard the rise in space temperature and delay the achievement of set-point. This would call for a longer pre-heat period — which incurs an energy penalty. At the end of occupation the heating would go off and any heat stored in the PCM would dissipate to no effect back into the unoccupied space, keeping it at an artifically elevated temperature and losing heat to outside at an unnecessarily-high rate until the PCM had resolidified.
Similar considerations apply to cooling. If the PCM is effective it will retard the effect of the room air-conditioning system. This could result in complaints, for example in settings such as hotels where this technology is being actively promoted.
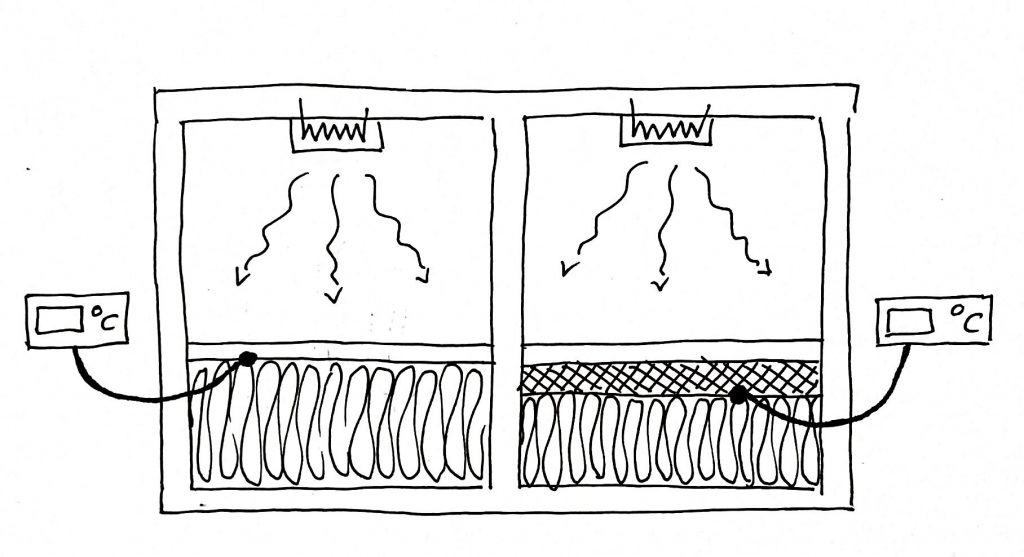
Skeptical of the online video purporting to demonstrate the effect (see diagram), I set up a simple mathematical simulation of the warm-up cycle of a heated room with PCM embedded behind plasterboard in its outer wall. The outcome was even worse than the description I gave earlier. At 5C outside the PCM did not start to store heat until the room temperature went nearly two degrees above the PCM melting point This is because the intervening plasterboard acts as a thermal insulator, keeping the PCM below the room temperature even though it I had 100mm of insulation on its cold side.
On a positive note, PCM layers may have a role to play in moderating overheating from solar gain to roofs in particular, for example in attic rooms. I have measured external roof-tile surface temperatures of 50C in the UK, which even with insulation behind it results in uncomfortable internal temperatures on the sloping ceiling behind it. 25mm of PCM under the roofing felt would absorb, by my calculations, the first 2.5 kWh per square metre of solar gain. Keeping the internal surface cooler would help alleviate discomfort and with internal insulation the stored heat would dissipate preferentially to the night sky.
For training on energy management topics see http://vesma.com/training